The department is headed by
Dr. Nagaraj Manickam, Ph.D, who
has been working in the field of kidney
disease for the past thirteen years including
six years of post-doctoral research
experience in the United States of America.
The Department of Vascular Biology at
MDRF was started in 2011, focusing on the
micro and macro vascular complications of
diabetes. The department aims at the
identification of novel early biomarkers and
associated signaling mechanisms in micro
and macro vascular complications under
differential glucose levels.
The department also provides training to
outside students in cell and molecular
biology techniques. A brief description of
the activities of the department is provided
below. |
Our vision is to identify early biomarkers as
well as a novel drug targets for diabetic
vascular complications. |
To execute experiments to identify early biomarkers for the progression of diabetic vascular complications.
To carry out experiments in vitro/in vivo models to unravel the molecular mechanisms involved in the pathogenesis of diabetic complications mainly chronic kidney disease and cardiovascular disease, thereby exposing a novel therapeutic drug target. |
Milestones and Key successes: |
I. Diabetic Kidney Diseases (DST Funded Project)
Diabetes is a global, life threatening and a devastating disease. Recently, International Diabetes Federation revealed that more than 463 million people globally have diabetes and these numbers are estimated to increase to 700 million by the year 2045. India has the second largest population of people with diabetes. The clinical importance of diabetes is that almost every organ of the body will get affected as the disease progress. The kidney is one of the major target organs, and nearly one third of the population with diabetes is at the risk of developing End Stage Renal Disease (ESRD). Diabetic kidney disease restricts a patient’s life physically, emotionally and socially since the disease makes its presence over every aspect of a person’s life style. Even if the perfect clinical regime is followed, diabetic kidney disease leads to either dialysis or kidney transplantation in many cases, thereby causing a burden on the individual and the society. Hence our department is focusing on the early detection of kidney injury induced by the metabolic syndrome. We have found that a molecule called asymmetric dimethyl arginine (ADMA) is elevated at an early stage of diabetic nephropathy and found a cut-off value for the same. In addition to this we have focused on the molecular mechanisms by which high glucose or ADMA causes kidney tissue injury so that we can find an effective drug target to prevent the
progression of diabetic kidney disease. Recently, we have dissected out a signaling pathway by which high glucose / ADMA causes kidney cell injury in terms of renal fibrosis. Below is the schematic representation of the signaling pathway.
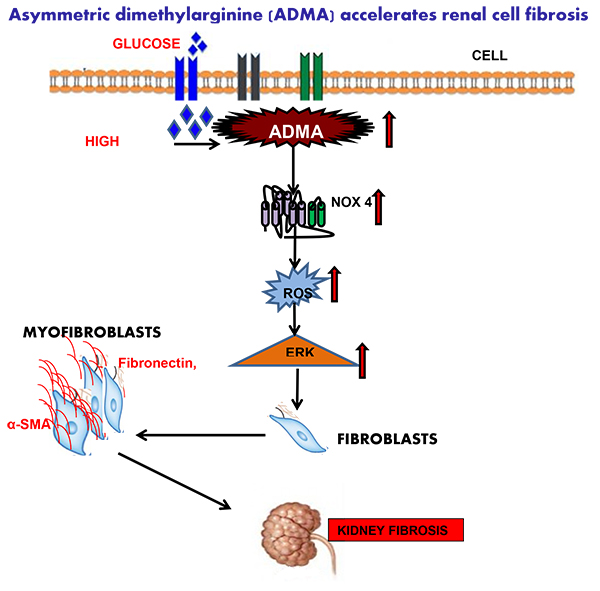
II. Role of Gasotransmitters and Treatment Strategies to combat DKD (DST-SERB funded Project):
In our previous study, we found that nitric oxide (a Gasotransmitter) was decreased upon ADMA accumulation. Recent studies are also pointing towards the role of other Gasotransmitters on the vasculature. Hence, we have extended our aim to study the role of Gasotransmitters in diabetic kidney disease and the regulation of Gasotransmitter metabolism. Hence, from the literature, we have picked up an Indian herbal plant (Aegle marmelos - AM) to study the protective role of its leaf extract and its active phytochemical components on diabetic kidney disease. Further, we are working on elucidating the possible molecular and cellular mechanism of AM in retarding the progression of nephropathy using diabetes induced C57BL/6 mice and in vitro models. We have explored and quantified the phytochemical contents such as alkaloids, flavonoids, phenolics, saponins, and tannin in the leaf extract. We also employed column chromatography, High Pressure Liquid Chromatography (HPLC) and GC-MS to explore the possible presence of active components in the leaf extract and found the presence of fifteen volatile bioactive components by analyzing with mass spectra of the constituents with the NIST library. Among these, we have selected two active components and as of now, experiments are going on to find the protective role of these components and leaf extract on the high glucose induced kidney cell injury using in vivo and in vitro models. We have found some very interesting results which indicate a promising effect of these active compounds in preventing the progression of diabetic kidney disease. Also, we are doing in-silico studies to analyse the molecular interactions and regulatory role of our novel compounds in high glucose mediated renal cell injury. These results will be validated with proper experiments.
III. Cardiovascular Disease (CVD) / Coronary Artery Disease (CAD) (DBT funded Project):
Our other aim is to look at the macro vascular complications of diabetes. Atherosclerosis is a leading cause of Cardiovascular disease in the metabolic syndrome. Hence, our focus is to find a novel biomarker for the disease and to find a drug target to impede the progression of the disease. After a great deal of experiments using different molecular biology techniques, we have found that a stress inducible protein called Sestrin2 has been significantly decreased on high glucose or OxLDL treatments in the monocytes and that the decrease in Sestrin2 leads to monocyte activation, trans-differentiation and foam cell formation (key events in the process of atherosclerosis) through the regulation of AMPK and mTOR. This study has recently been published in a high impact journal. It is the first of its kind in dissecting out a molecular signaling pathway involving sestrin2-mTOR under high glucose and dyslipidemic conditions. We have arrived at the novel cell signaling pathway as shown below
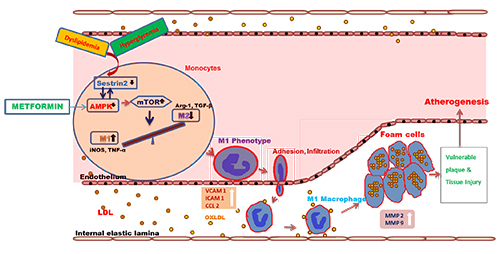
Further, we have found the status of Sestrin2 in the monocytes and circulation of people with/without diabetes and/or dyslipidemia. We have found a significant association and correlation of sestrin2 levels with lipid parameters.
Further to understand the role of Sestrin2, we have utilized animal models and found that the sestrin2 expression levels were decreased in the aorta and heart tissues of Wistar rats.
Status of all isoforms of sestrins and its role in ER stress and Oxidative stress under diabetes and dyslipidemic conditions
After finding a significant decrease in the levels of Sestrin2, we were interested in looking at all isoforms of sestrins. Also, we were interested in studying the role of sestrins in ER stress as sestrin2 regulates mTOR. Hence, in this study, we have studied the status of all isoforms of sestrins and the status of ER stress markers and their association with sestrins.
1. Sestrin1 mRNA expression of monocytes was significantly decreased in individuals with dyslipidemia and diabetes with dyslipidemia while compared with controls. Sestrin3 was a significantly decreased in dyslipidemia while compared with control groups.
2. Sestrin1 negatively correlated with glucose tolerance, triglyceride, BMI and positively correlated with HDL. Sestrin3 had significant negative correlation with BMI, triglyceride and plasma atherogenic index.
3. mRNA expressions of ER stress markers in PBMC were increased in individuals with dyslipidemia and diabetes with dyslipidemia when compared to controls. mRNA expression of ER stress markers of PBMC positively correlated with BMI, FBS, PBSS, HbA1c, HOMA-IR, triglycerides and negatively with HDL-C and Sestrin2 expression in monocytes and serum.
Sestrin2 regulates the ER stress markers and oxidative stress markers expression in THP1 cells. Over expression of Sestrin2 reduced the effect of high glucose on ER stress markers expression.
IV. MicroRNAs/Sestrin2/Atherosclerosis/Diabetes Relationship (ICMR Funded Project):
In our previous study we have identified and proved that Sestrin2 counteracts monocyte activation under hyperglycemic and dyslipidemic conditions. Further, our studies point out that there exists impaired sestrin2 and its downstream signaling in diabetes and its cardiovascular consequences. But the reason for the decrease in the sestrin2 expression under stress condition such as hyperglycemic/hyperlipidemic states is not clearly understood. Recent studies imply a role of miRNAs in altering gene expression and biological activities. Therefore, targeting miRNAs has recently been conceived as a potential strategy to beneficially modulate the biomarkers. Hence in this study, we are investigating the circulatory/monocyte levels of miRNAs specific to sestrin2 gene in T2DM patients with/without atherogenic plaques. Our second objective is to target and study the role of those specific miRNAs in regulating sestrin2 expression and monocyte activation under in vitro conditions.
Training Programs at our Department
Our department is giving laboratory training programs for the B.Tech and M.Tech students |
S. No
|
Title |
Funding Agency |
Period |
| 1. |
Asymmetric dimethylarginine (ADMA) in diabetic nephropathy and its role in hyperglycemia mediated stress signaling in kidney cells.
(Principal Investigator) |
DST |
2013-2017 |
| 2. |
Exploring a novel biomarker and drug target: Role of Sestrin, a TOR (Target of Rapamycin) inhibitor, in diabetic dyslipidemia.
(Principal-Investigator) |
DBT |
2013-2017 |
|
S. No
|
Title |
Funding Agency |
Period |
| 1. |
Exploring the mechanistic role of Aegle marmelos in the prevention of diabetic nephropathy: Involvement of Gasotransmitters in the regulation.
(Principal Investigator) |
DST-SERB |
2017-2020 |
| 2. |
Investigating the miRNA/Sestrin2 relationship in type 2 diabetes patients with and without atherosclerosis & targeting miRNAs for altering monocyte activation/pro-atherogenic cascades (Principal Investigator) |
ICMR |
2019-2022 |
|